Dr. Mudassar Altaf, Associate Professor of Chemistry, Department of Higher Education, Government of the Punjab, Pakistan
Contents:
- Definition and examples of oxides
- Types of oxides
- Acidic oxides: definition, examples and characteristic features
- Basic oxides: definition, examples and characteristic features
- Amphoteric oxides: definition, examples and characteristic features
Oxides:
Definition: “The chemical compounds containing at least one oxygen atom chemically bonded with metallic or non-metallic atom are called oxides”. For example, carbon monoxide (CO), carbon dioxide (CO2), calcium oxide (quicklime or burnt lime: CaO) etc.
Types of Oxides:
These are classified into basic, acidic and amphoteric oxides; as discussed below.
Acidic Oxides:
Definition: “The non-metallic oxides that react with water to produce acids are called acidic oxides”. For example, carbon monoxide (CO), carbon dioxide (CO2), sulphur dioxide (SO2), nitrogen dioxide (NO2) etc.
Characteristics of Acidic Oxides:
- They are non-metallic oxides. For example, C, S and N are non-metals in CO, CO2, SO2 and NO2.
- They are soluble in water and increase acidity of the solution by lowering the pH value. These oxides react chemically with water and form acids. Hence, these are called acidic oxides. CO2 is a colourless gas and soluble in water at 25°C by 0.145g per 100 cm3 of water. SO2 is also colourless gas and soluble in water at 25°C by 9.4g per 100 cm3 of water.
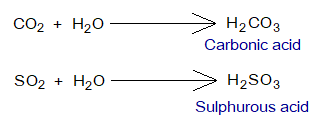
- Acidic oxides react chemically with alkalis to produce salt and water. Go through the following examples.
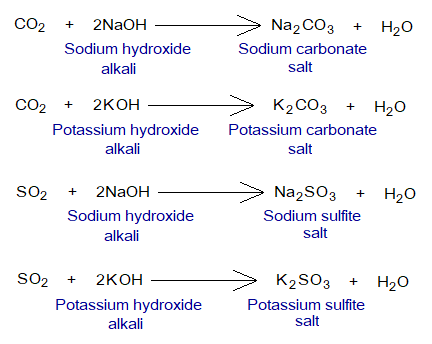
The hydrogen-carbonates are also formed if limited amount of alkali is used instead of excess. For example, NaHCO3, KHCO3, NaHSO3, KHSO3 sodium hydrogen-carbonates, potassium hydrogen-carbonate, sodium hydrogen sulfite, potassium hydrogen sulfite respectively. The hydrogen-carbonates are also called bicarbonates, like sodium bicarbonates, potassium bicarbonate for the given examples. Moreover, the ‘sulfite’ can also be written as ‘sulphite’.
Basic Oxides:
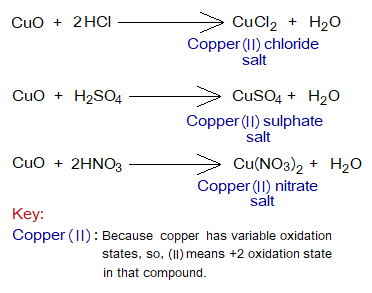
Definition: “The metallic oxides that react with water to produce alkalis are called basic oxides”. For example, calcium oxide (CaO), copper oxide (CuO) etc.
Characteristics of Basic Oxides:
- They are metallic oxides. For example, Ca, and Cu are metals in CaO, & CuO.
- Mostly, they are insoluble in water, like CuO (a black solid powder). But CaO reacts with water to form Ca(OH)2. CaO is a white or light yellowish powder and readily reacts with water to show exothermic reaction and the mixture becomes so hot. Slake is derived from an old English word ‘slacian’ meaning ‘having thirst of water’; because of violent reaction with water.
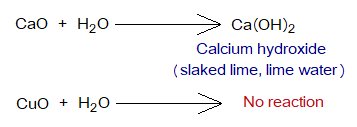
Metallic oxides react with acids to produce salts and water, called neutralization reactions.
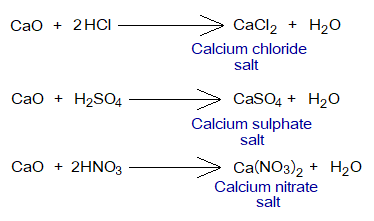
Amphoteric Oxides:
Definition: “The oxides that can behave both ways, as an acid and a base are called amphoteric oxides”. The ‘ampho’ is a Greek word, meaning ‘both’. Among such oxides, aluminium oxide (Al2O3), and zinc oxide (ZnO) etc. are examples.
Characteristics of Amphoteric Oxides:
- These oxides react with acids and bases. They behave as a base when react with an acid; and vice versa.
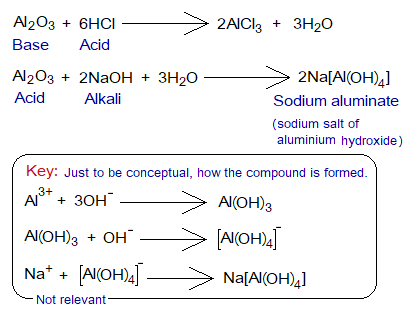
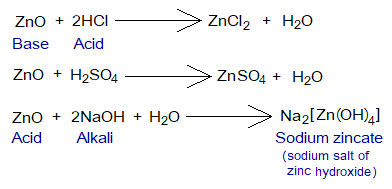
- Another feature is that non-metallic as well as metallic oxides can show amphoteric nature. In above examples, Al and Zn both are metals. However, boron trioxide (B2O3), lead(II) oxide (PbO), are the examples where boron is non-metal, while lead is a metalloid (semimetal).
Exercise:
Like Na[Al(OH)4], try to explain formation of Na2[Zn(OH)4] that how the valencies are satisfied in this compound. Oxidation state of Zn is +2.



