Dr. Mudassar Altaf, Associate Professor of Chemistry, Department of Higher Education, Government of the Punjab, Pakistan
Contents:
- Definition of acids and examples
- Definition of acidic hydrogen
- Definition of alkalis and examples
- Etymology of alkali
- Proton donor versus acceptor
- Neutralization reaction
- Bases versus alkalis
- Chemical reactivity of acids: Reactions with metals, bases, carbonates, & bicarbonates
- Chemical reactivity of bases: Reactions with acids, & ammonium salts
- Acid base indicators: litmus, thymolphthalein, methyl orange, universal indicator
- Strong versus weak acids: Definition & examples
- Bow tie diagram and pH-scale
- Carboxylic acids – a brief introduction
- Mineral acids – a brief introduction
- Definition of basicity in terms of alkalinity
Acids: Definition
The acids can be defined as “the chemicals that donate hydrogen ions (H+) in aqueous solutions”. For example, hydrochloric acid (HCl), nitric acid (HNO3), acetic acid (CH3COOH), sulfurous acid (H3SO3), sulfuric acid (H2SO4), phosphorous acid (H3PO3), phosphoric acid (H3PO4) etc.
Acetic acid (aka ethanoic acid) molecule contains two types of hydrogen atoms. In the following diagram, the red hydrogen is the only acidic hydrogen – the hydrogen that is released as proton (H+) when the substance shows acidic properties in aqueous solution. It is the hydrogen that defines the solution the acidic one.
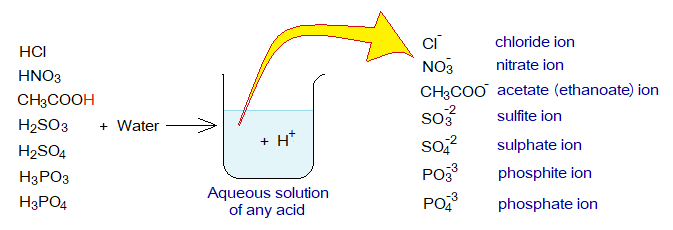
Exercise 1:
Write balanced chemical equations of above acids with water to produce hydrogen ions and anions.
Alkalis: Definition
The alkalis are bases and carry hydroxide ion (OH–). All alkalis are bases, but all bases are not alkalis. Alkali can be defined as “the chemicals that donate hydroxide ion (OH–) in aqueous solutions”. For example, sodium hydroxide (NaOH), potassium hydroxide (KOH), ammonium hydroxide (NH4OH), calcium hydroxide [Ca(OH)2], magnesium hydroxide [Mg(OH)2], aluminum hydroxide [Al(OH)3] etc.
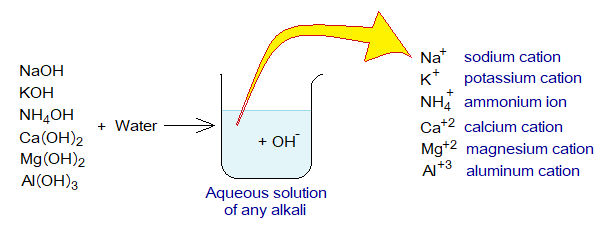
Exercise 2:
Write balanced chemical equations of above alkalis with water to produce hydroxide ions and cations.
The etymology of alkali is Arabic from the words “al” meaning “the” and “qali” meaning “ashes”. Because, the ancient people, used ashes for making soap and glass. The ash was obtained by burning plants; and that contains alkalis of soda and potash.
Proton Donor Versus Acceptor:
- Acids are proton donors; while alkalis (bases) are proton acceptors.
- When acid and base react chemically, the neutralization reaction takes place. As a result of this reactivity, salt and water are formed.
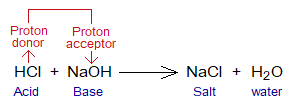
- The anion of an acid makes ionic bond with cation of the base to produce salt.
- The neutralization is the chemical combination of H+ and OH– ions to produce water. On the pH scale. the pH 7 is neutral; while 0 to 6 is acidic, and 8 to 14 is basic. So, water makes the pH neutral. In other words, hydrogen & hydroxide ions balance each other.
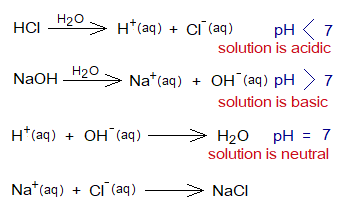

Bases Versus Alkalis:
- Base is a general term, including all the types of bases and alkalis. For example, metallic oxides are also bases; like, sodium oxide (Na2O), sodium peroxide (Na2O2), potassium oxide (K2O), calcium oxide (CaO), magnesium oxide (MgO), zinc oxide (ZnO), iron(II) oxide (FeO), iron(III) oxide (Fe2O3).
- The metallic hydroxides are also bases. For example, sodium hydroxide (NaOH). The few other examples are given above. These bases are classified as alkalis.
- All alkalis are soluble in water. They produce hydroxide ion (OH–) when dissolved in water.
- The soluble metallic oxide bases form hydroxide when dissolved in water.
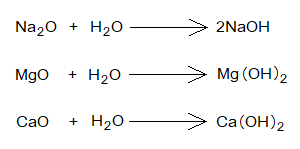
- The FeO, Fe2O3 and CuO are the metallic oxides that are insoluble in water.
Chemical Reactivity of Acids:
It is a characteristic property of the acids that they react chemically with metals, bases and carbohydrates.
- Reactions with Metals: The acids produce hydrogen gas and salt when react with metals. In the following examples of iron, zinc and sodium metals, the ferrous sulphate, zinc chloride and sodium chloride respectively the salts are formed along with hydrogen gas.
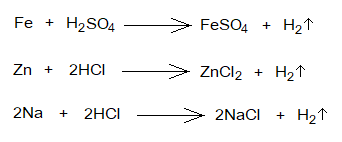
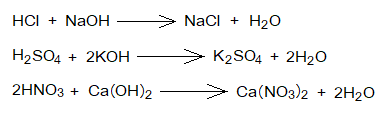
- Reactions with Bases: Acids and bases react chemically together to produce salt and water, the reaction is called neuralization, as described above. Sodium chloride, potassium sulphate and calcium nitrate salts are formed along with water by the following examples of chemical reactions between acids and alkalis.
The metallic oxides also react with acids to produce salts and water. For example, CaO reacts with HCl to from CaCl2 & H2O. It forms Ca(NO3)2 when reacts with HNO3. Similarly, MgO, Fe2O3 show chemical reactivity with acids to produce salts and water.
- Reactions with Carbonates: Acids react chemically with metal carbonates to produce salts, carbon dioxide gas and water.
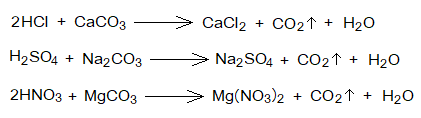
- Reactions with Bicarbonates: Like carbonates, acids also react chemically with bicarbonates (hydrogen carbonates, baking soda) to form salts, carbondioxide gas and water.
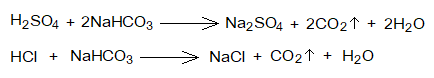
Chemical Reactivity of Bases:
It is a characteristic property of bases that they react chemically with acids and ammonium salts.
- Reactions with Acids: Alkalis react with acids to produce salt and water, the neuralization reaction has been discussed above.
- Reaction with Ammonium Salt: The alkalis react with ammonium salts to produce salts, water and ammonia gas.
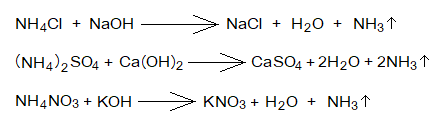
Acid base indicators:
Definition: “The substances that change their own colour in acidic or basic medium at particular pH level are used to test the pH, i.e., the acidity, neutrality or alkalinity of a solution”. Litmus, thymolphthalein, methyl orange and universal indicators have been discussed here under in this context.
- Acid base effect on litmus
- Acid base effect on thymolphthalein
- Acid base effect on methyl orange
- Universal indicator
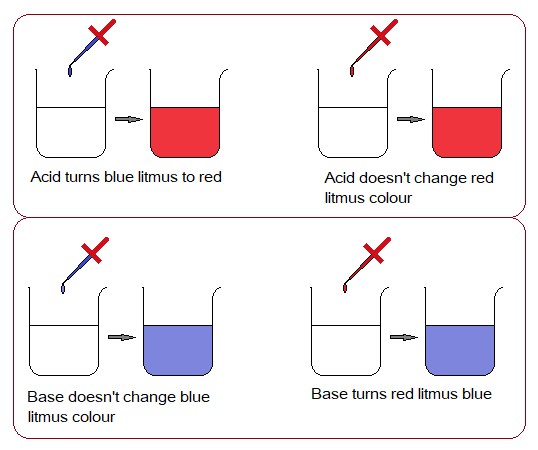
1. Acid Base Effect on Litmus:
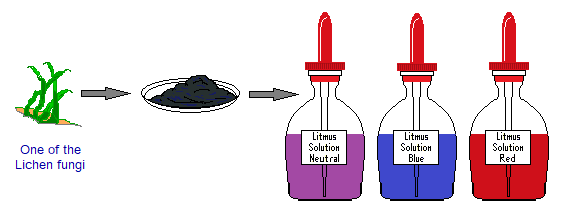
Litmus is a powder dye of black colour with bluish tone (onyx blue) that can easily be dissolved in distill water to give purple neutral solution. It changes its colour in acidic or basic media; so, used as an indicator to know what the nature of the solution is. It gives red colour in acidic solution and turns blue in basic media.
By etymology, litmus is derived from old Scandinavian language. The “litr” meaning “colour” and “mosi” is for “moss” plants. A dye (colour) is extracted from the lichen plant (considered a type of moss; actually, a fungi). The leaves are dried and dissolved in distil water to make a neutral solution of litmus. For blue litmus, the aqueous solution is made in basic medium; and for red litmus, the aqueous solution is made in acidic medium.
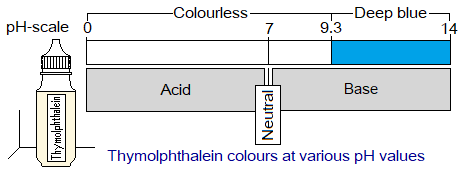
2. Acid Base Effect on Thymolphthalein:
It is synthetic acid-base indicator and doesn’t exist naturally. It is a white powder but its solution is prepared to work as indicator. The indicator is colourless in acid and neutral solution; while turns to deep blue in alkaline solution.
The pH of an acid is 0 to 6.9; 7.0 is neutral; while 7.1 to 14 is basic. Because, it shows colourless below pH 9.3 to 0, thus doesn’t tell the exact nature of the solution whether it is acidic or neutral, or might be slightly basic. Its deep blue colour appears around 9.3 and stays to 14, a typical range of pH for basic solutions.
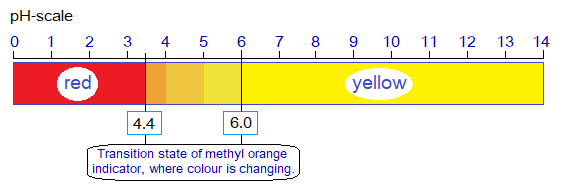
3. Acid Base Effect on Methyl Orange:
Like thymolphthalein, methyl orange is also man-made acid-base indicator. It is yellow or orange solid and having solubility in water. It shows a red colour in an acidic medium in the range of PH 0 to 4.4; it turns yellow above this range and stays on this colour to the end of pH scale. So, weak acids, neutral, and weak or strong bases all show yellow colour with methyl orange indicator; and thus, discrimination is an issue. Only strong acid solutions can be identified. In a true sense, it changes its colour in the range of pH 4.4 to 6.0.
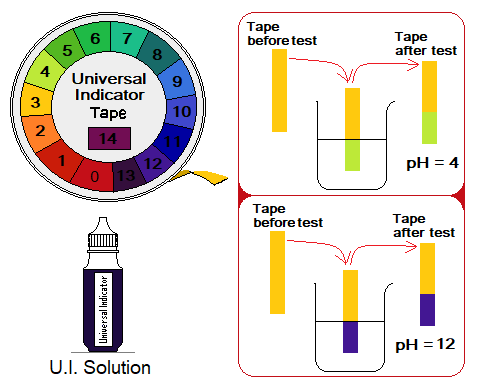
4. Universal Indicator:
The composition of a universal indicator is a combination of several indicators, e.g., phenolphthalein, bromothymol blue, thymol blue, methyl red, methyl orange. The universal indicator covers entire pH-scale by showing different colours. When a drop of indicator is added into a solution under examination, the colour appeared is matched with the colour scheme provided which will tell the pH value.
The universal indicator is available in two forms; (1) solution form. (2) paper form.
Strong versus Weak Acids:
All acids do not dissociate completely in aqueous solution to liberate acidic hydrogen. The extent of dissociation varies among acids.
Strong Acids: These can be defined as “the acids that are completely ionized into aqueous solution”. They show their pH low. For example:
- Hydrochloric acid (HCl): dissociates into H+(aq) & Cl–(aq) ions
- Hydrobromic acid (HBr): dissociates into H+(aq) & Br–(aq) ions
- Hydroiodic acid (HI): dissociates into H+(aq) & I–(aq) ions
- Nitric acid (HNO3): dissociates into H+(aq) & NO3–(aq) ions
- Perchloric acid (HClO4): dissociates into H+(aq) & ClO4–(aq) ions
- Sulphuric acid (H2SO4): dissociates into H+(aq) & HSO4–(aq)
Cl–, Br–, I–, NO3–, ClO4– & HSO4– are called chloride, bromide, iodide, nitrate, perchlorate, & bisulphate (hydrogen sulfate) ions respectively.
Weak Acids: These can be defined as “the acids that are ionized partially into aqueous solution”. They show their pH 4 to 6.9. For example:
- Acetic acid (CH3COOH): dissociates into H+(aq) & CH3COO–(aq) ions
- Carbonic acid (H2CO3): dissociates into H+(aq) & CO32-(aq) ions
- Formic acid (HCOOH): dissociates into H+(aq) & HCOO–(aq) ions
- Oxalic acid [(COOH)2]: dissociates into H+(aq) & COO– (aq) ions
- Benzoic acid (C6H5COOH): dissociates into H+(aq) & C6H5COO– (aq) ions
- Phosphoric acid (H3PO4): dissociates into H+(aq) & PO4-(aq) ions
- Hydrofluoric acid (HF): dissociates into H+(aq) & F–(aq) ions
CH3COO–, CO32-, HCOO–, COO–, C6H5COO–, PO4-, & F– are called acetate, carbonate, formate, oxalate, benzoate, phosphate and fluoride ions respectively.
On the pH scale, these can be expressed by the bow tie diagram. This diagram, shows strong & weak acids, neutral solutions, and weak & strong bases. Moving towards 7, gradually the strength of acidity decreases. In other words, ability to donate proton (H+) decreases.
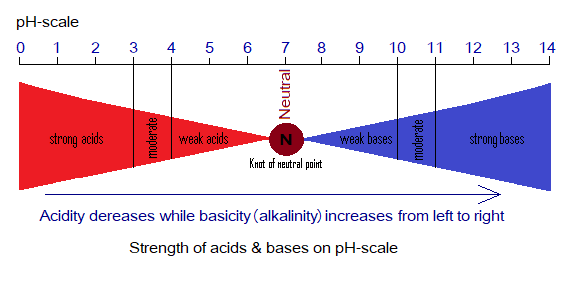
Carboxylic Acids: A Brief Introduction
“The organic acids containing carboxylic group (-COOH) are called carboxylic acids”. In old days, their source was organic material only; so, are recognized by this term the ‘organic acids’; for example, acetic acid (ethanoic acid CH3COOH). The household vinegar is about 5% acetic acid solution. Nowadays, carboxylic acids are made synthetically.
Mineral Acids: A Brief Introduction
“The inorganic acids are called mineral acids”. In earlier times, their source was minerals; so, they are recognized by this term; for example, HCl, HNO3, H2SO4 etc. Nowadays, these are made synthetically.
Exercise 3:
Remember the names of the carboxylic acids and their formulae given above.
Exercise 4:
Construct the ionic equations for strong and weak acids’ dissociation in water.
Exercise 5:
Describe basicity (alkalinity) on pH-scale by using the above bow tie diagram. “In terms of alkalinity, the basicity is the strength (ability) of a base to release hydroxide ions (OH–) in aqueous solution”.



