Dr. Mudassar Altaf, Associate Professor of Chemistry, Department of Higher Education, Government of the Punjab, Pakistan
Contents:
- Salts’ various definitions
- Salts’ preparation:
- Titrations between acids & alkalis
- Metals & acids reactions
- Insoluble bases & acids reactions
- Insoluble carbonates & acids reactions
- Solubility definition and rules of salts’ solubility
- Solubility of alkalis
- Hydrated versus anhydrous substances
- Water of crystallization
- Reversibility between anhydrous & hydrated substances
Salts:
Definitions: There are various definitions of the salt, as under:
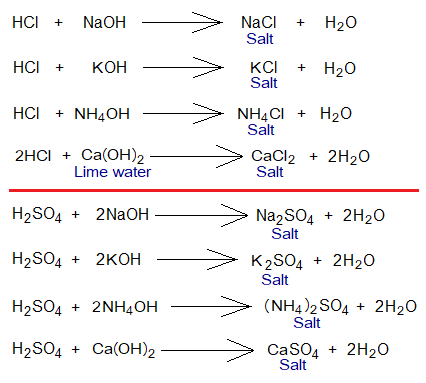
- “The compound that is formed by electrostatic forces of attraction between metallic cation or a few other non-metallic cations and non-metallic anion”. The best-known non-metallic cation is ammonium ion (NH4+); the K+ is a metallic cation, and Cl– (chloride)is non-metallic anion; both establish ionic bond due to carrying opposite charges; so, NH4Cl and KCl salts are formed.
- “The H+ of an acid is replaced by a metallic cation or a few other non-metallic cations to form an ionic compound that is called a salt”. For example, HCl is an acid, which replaces its acidic hydrogen with Na+, then, NaCl salt is formed.
- “The neutralization reaction between an acid and base forms an ionic compound called salt”. The second product of this reaction is water. For example, HCl and NaOH react chemically to form NaCl salt and H2O.
Salts’ Preparation:
Following reactions have been discussed here.
- Titrations between Acids & Alkalis: In-detail titration has been discussed on the link https://chemiologist.com/titration-technique-in-chemistry/ . The aqueous solution of an acid of certain molarity is taken in the burette and drop-by-drop allowed to fall in titration flask containing aqueous solution of an alkali and the indicator. The titration flask usually contains 10 or 25 cm3 of alkali solution and 1 or 2 drops of phenolphthalein indicator. This indicator shows pink colour in alkali solution; and on an equivalence point (end point) its colour fades almost, say just to colourless on adding one more drop of an acid. The end point is the point where the reaction between acid and the base is accomplished. Consequently, salt and water are formed. Titration is a quantitative analysis technique and is not used to make salts.
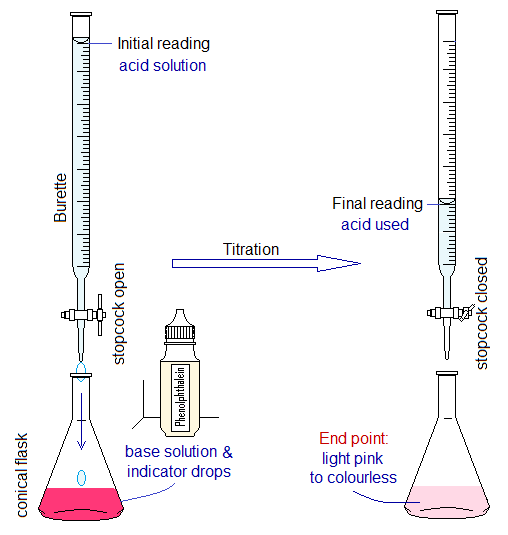
Usually, the solutions are made having their molarities 0.1, 0.05 molar etc.
- Metals & Acids Reactions: The salts can also be prepared by the reaction of a metal with an acid. Hydrogen gas is another product of these reactions. The metal is used in excess in order to make sure that all acid has been consumed.
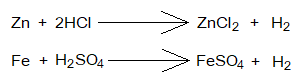
These are Redox reactions. For example, the HCl dissociates into its ions, H+ & Cl–. Metal undergoes into oxidation [Zn=Zn+2+2e–]. Hydrogen ions show reduction and ultimately form hydrogen gas molecule [2H++2e–=2H=H2].
Zinc chloride and ferrous sulfate are highly soluble salts in water at room temperature, and their solubility increases with rise in temperature. Their solubility is as under:
ZnCl2 = 432g/100 cm3 at 25°C
FeSO4 = 30g/100 cm3 at 25°C
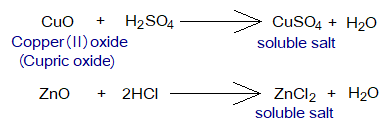
- Insoluble Bases & Acids Reactions: When water-insoluble bases like zinc oxide (white powder) or copper oxide (black powder) react chemically with acids, the soluble salts and water are formed. Step-by-step procedure is given below:
- Take dilute acid in a beaker.
- Add insoluble base in excess, but pinch by pinch, while doing a constant stirring.
- Warm the solution on the flame if needed to speed up the reaction.
- The base reacts with acid and the soluble salt is formed along with water. So, the all the base is consumed and disappeared initially.
- But on adding more and more base, when it starts going to settle down instead of disappearing, it means that all the acid has been consumed. Stop adding more base.
- Let the solution to cool at room temperature. Filter the unreacted base.
- The filtrate is heated to evaporate, so to get the crystals of soluble salt on the bottom of the beaker.
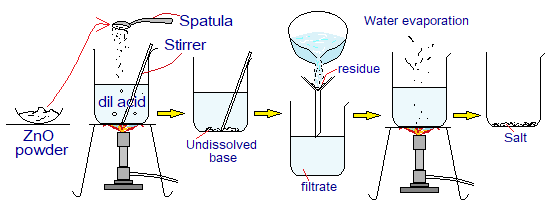
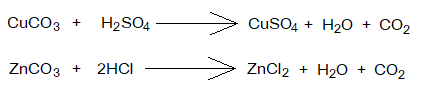
- Insoluble Carbonates & Acids Reactions: When water-insoluble carbonates like copper carbonate or zinc carbonate react chemically with acids, the soluble salts, carbondioxide gas and water are formed. Step-by-step procedure is given below:
- Take dilute acid in a beaker.
- Add insoluble carbonate salt in excess, but pinch by pinch, while doing a constant stirring.
- Effervescence occurs because of formation of CO2 gas; that releases out and bubbles are formed. Effervescence has its Latin origin from the word ‘effervescere’ meaning ‘making bubbles on boiling’. It is a rapid escape of gas from the liquid, like CO2 releases from fizzy drinks when a pinch of salt is added.
- When bubbling stops on adding more carbonate, stop adding more, Because, the acid has been consumed fully.
- Filter the unreacted salt of carbonate. The filtrate contains yield of water and soluble salt; while the carbondioxide gas has been released.
- The filtrate is heated to evaporate, so to get the crystals of soluble salt on the bottom of the beaker.
Solubility Definition & Rules of Salts’ Solubility:
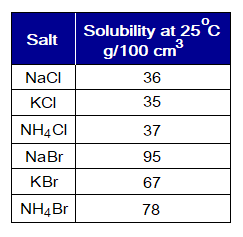
Definition: The term solubility can be stated as, “the maximum amount of a solute that can be dissolved in a given amount of the solvent at a specified temperature”. Usually, the solubility is measured in grams per 100 cm3 or grams in 1 dm3 at 25°C (room temperature).
- The salts of Na+, K+ and NH4+ are soluble in water. For example,
- All salts of nitrates are soluble in water. For example,
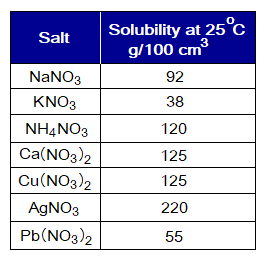
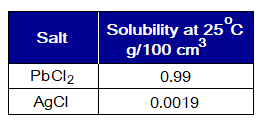
- Except chlorides of lead (Pb2+) and silver (Ag+), all others are soluble in water. The salts, PbCl2 and AgCl, are very poorly soluble.
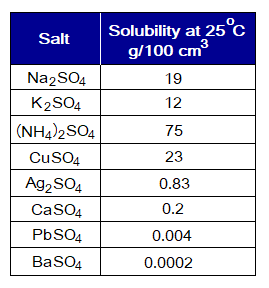
- All salts of sulphates are soluble in water, except those of Ag+, Ca2+, Pb2+ and barium (Ba2+) having extremely low.
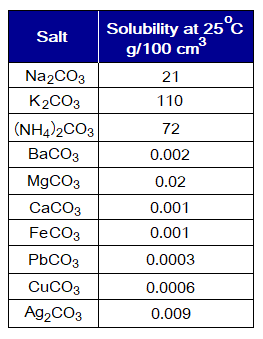
- The carbonates of alkali metals (I-A) are highly soluble in water, examples, Na+, K+ carbonates. The NH4+ carbonate salt is also highly soluble. However, the carbonates of alkaline earth metals (II-A) are insoluble or sparingly (in minute quantity) soluble, examples, Ba2+, Mg2+, Ca+2 carbonates. In general, except the carbonate salts of Na+, K+ and NH4+, all others are insoluble.
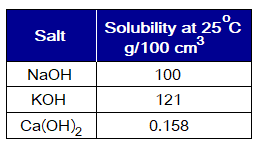
Solubility of Alkalis:
The hydroxides of alkali metals Na+, K+ and NH4+ highly soluble in water at room temperature. However, the hydoxide of Ca2+ is partially (sparingly) soluble. All other hydroxides are insoluble.
- NaOH (caustic soda, lye) and KOH (caustic potash) are a white inorganic crystalline solid compounds with the appearance of deliquescent chips. “The substances that absorb moisture from atmosphere and dissolve in absorbed water are called deliquescent compounds”. The ‘deliquescent’ is a Latin word meaning ‘dissolving’.
- Ca(OH)2 (slaked lime) is a white powder inorganic compound. Its solubility decreases with rise in temperature and vice versa, called retrograde (inverse) solubility. “The solubility of a substance that decreases with rise in temperature, is called inverse solubility”.

Hydrated Substances:
Definition: “The substances that contain a certain number of water molecules chemically combined with the molecule of that substance, are known as hydrated substances”. For example:
- Plaster of Paris (calcium sulphate hemihydrate), CaSO4. ½H2O; a white powder in its appearance. One molecule of compound contains half molecule of water, so called hemihydrated.
- Copper (II) sulphate pentahydrate, CuSO4.5H2O; a bright sky-blue powder in its appearance.
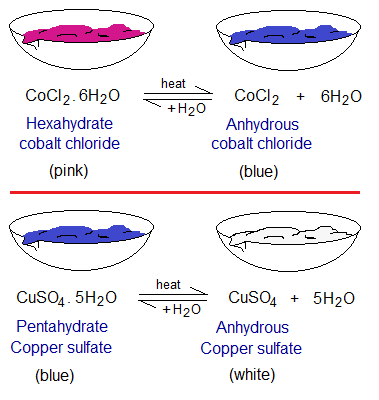
- Cobalt chloride hexahydrate, CoCl2.6H2O; a pink powder in its appearance.
- Washing soda is a hydrated salt, Na2CO3.10H2O. It is white powder of sodium carbonate decahydrate. There are 10 water molecules making it hydrated, so, recognized as ‘decahydrate’.
Water of Crystallization: “The water molecule as a part of a crystalline structure, making the substance hydrated is called water of crystallization”. In the above examples, ½H2O, 5H2O and 6H2O. These are written after chemical formula and having a dot in-between.
Hemi…, mono…, di…, tri…, tetra…, penta…, hexa…, hepta…, octa…, nona…, and decahydrate are used for ½, 1, 2, 3, 4, 5, 6, 7, 8, 9, and 10 water of crystallization respectively.
Anhydrous Substances:
Definition: “The substances that do not contain water of crystallization in their structure, are known as anhydrous substances”. For example:
- Baking soda: sodium hydrogen-carbonate, NaHCO3; a white powder in its appearance. Also called sodium bicarbonate.
- Anhydrous copper sulphate, CuSO4; a white powder in its appearance.
- Anhydrous cobalt chloride, CoCl2; a blue powder in its appearance.
Reversibility between Anhydrous & Hydrated Substances:
In-detail discussion has been given on the link https://chemiologist.com/chemistry-of-reversible-reactions-and-their-industrial-application/ . Go through the concept related to the examples:
- CuSO4.5H2O into CuSO4; and reverse.
- CoCl2.6H2O into CoCl2; and reverse.