Dr. Mudassar Altaf, Associate Professor, Higher Education Department, Government of the Punjab, Pakistan
Definition:
The organic compounds containing methylene group (-CH2-) that is chemically reactive by containing two acidic hydrogens due to having bonding with two electron withdrawing groups are called active methylene compounds.
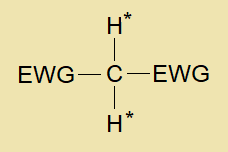
In the figure, EWG stands for ‘electron withdrawing group’; the central carbon is active methylene containing two acidic H* atoms.
Reactivity:
The base attacks on acidic hydrogen and creates a negative charge on the carbon atom (carbanion) of methylene by removing H+ ion. The carbanion is a reactive conjugate base and plays its role as active methylene in the compound. The (H+) cation of hydrogen also called ‘hydron’ or ‘proton’ makes water with hydroxide (OH–) of an alkali.
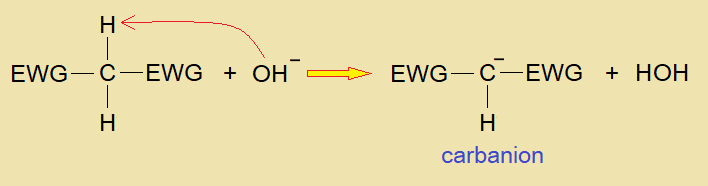
Electron Withdrawing Group:
It is defined as the atom or a group of atoms that can pull (draw or drag) the electron density towards itself from neighboring atoms and attains a partial negative charge on itself by pi (π) bonds through inductive, resonance (mesomeric) or electromeric effect. It is denoted by (-M). Inductive effect is simply related with induction that means to induce (bring about) the partial charges due to electron withdrawing or electron donating group due to difference in electronegativity (E.N.) values; this is a simple pull or push of shared pair of electrons. The E.N. of carbon is 2.5; and that of hydrogen is 2.2. So, relatively, the larger E.N. value of carbon makes hydrogen an acidic hydrogen, carrying partial positive charge and produces hydron during chemical reaction. Resultantly, the partial negative charge on carbon makes carbanion. The inductive effect is observed in sigma bonds.
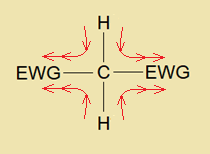
The mesomeric effect also called the resonance effect, is the polarity induced due to pi-pi overlapping. The mesomeric effect brings structural change in contributing structures (mesomers). The electromeric effect occurs when an attacking reagent is either nucleophile (Nu–) or an electrophile (E+) that attacks on a molecule and pi-bond electron shifts so that to create negative or positive charge on the molecule. The electromeric effect is observed in pi-bonds. Few examples of electron withdrawing groups are:
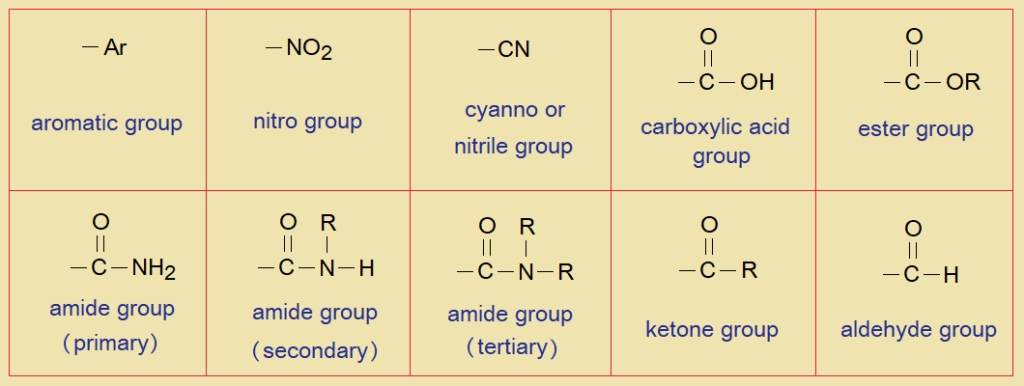
Following are few examples of chemical reactions where active methylene group is involved in their mechanism:
- Acylation: https:/acylation-between-n-tert-butoxycarbonyl-l-phenylalanine-and-malononitrile-mechanism-of-chemical-change/
- Aldol condensation: https:/aldol-condensation/
- Alkylation: https:/alkylation-of-3-oxobutanenitrile-with-methanol-mechanism-of-chemical-change/
- Arylation: https:/arylation-of-malononitrile-with-4-bromotoluene-mechanism-of-chemical-change/
- Claisen condensation: https:/claisen-condensation-reaction-mechanism-of-chemical-change/
- Claisen Schmidt condensation: https:/condensation-between-acetone-benzaldehyde-into-benzylidene-acetone-claisen-schmidt-mechanism/
- Darzens glycidic ester synthesis: https:/darzens-glycidic-ester-synthesis/
- Knoevenagel condensation: https:/knoevenagel-condensation-reaction/
- Ketone self-condensation: https:/self-condensation-in-ketones/
- Mannich reaction: https:/mannich-condensation-reaction-for-the-synthesis-of-beta-amino-carbonyl-compound-a-mannich-base/
- Perkin condensation: https:/perkin-condensation-synthesis-of-cinnamic-acid/
- Reformatsky condensation: https:/reformatsky-condensation-reaction-synthesis-of-s-hydroxyester/
- Stobbes condensation: https:/stobbes-condensation-reaction/
- Wittig reaction: https:/wittig-reaction-synthesis-of-alkene-with-ylide/



