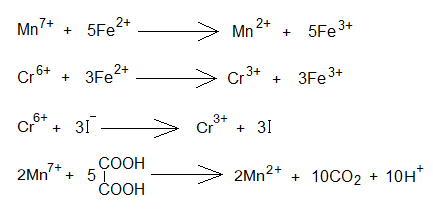Dr. Mudassar Altaf, Associate Professor of Chemistry, Department of Higher Education, Government of the Punjab, Pakistan
Contents:
- Oxidation number (oxidation state): Definition
- Various aspects of oxidation state
- Use of Roman numerals
- Redox: The term
- Definitions of reduction & oxidation
- Redox reaction of KMnO4 with H2S
- Redox reaction of KMnO4 with H2C2O4
- Redox reactions of aqueous KI with K2Cr2O7
- Oxidizing agent versus reducing agent: Definitions
Oxidation Number:
Definition: “it describes the role of an atom in a compound that how much that atom gains, loses or shares electron(s) in a chemical bond.” It is also called ‘oxidation state’; and is a hypothetical term based upon mathematical calculations to describe the charge on an atom in a compound. Further, the electronegativity difference also describes the nature of the bond either pure ionic, ionic-covalent or pure covalent. For example:
- H2 is a pure covalent bond, no one atom is ionic, but both are sharing 1 electron to fulfill their need to attain noble gas electronic configuration. Think in terms of electronegativity difference (∆E.N.) why it is pure covalent?
- HCl is an ionic-covalent bond, no one atom is purely ionic, but the role is hypothetical and can be described as +1 of H & -1 of Cl in terms of oxidation states. Because of ∆E.N. between two atoms, a partial positive charge is created on hydrogen (Hᵟ+) while partial negative on chlorine (Clᵟ–). By Pauling scale the value of electronegativity of H is 2.20, Cl is 3.16; and the difference is 0.96. Partial negative charge is created on the atom having relatively the greater value, and vice versa. Think why chlorine attains partial negative, while hydrogen partial positive?
- NaCl is a pure ionic bond. Oxidation state of sodium is +1 by sodium cation & chlorine -1 by chloride anion.
Various Aspects of Oxidation State:
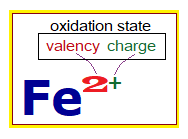
- The oxidation state is a combination of valency and charge. Valency is the combining capacity of an atom; and the charge is either positive or negative due to lose or gain of electron(s).
- The neutral atoms of an element have their oxidation state ‘zero’. In other words, it is neutral by having equal number of protons and electrons. For example, Lithium (Li) having its oxidation number ‘zero’ is due to 3 electrons and 3 protons. Similarly, oxygen atom is neutral by its 8 electrons (atomic number 8, as proton number 8). If the number of electrons is same to the atomic number (sum of proton number), then the atom would be neutral as a whole.
- The compound is overall neutral due to equal number of negative and positive oxidation states. For example, NaCl by Na+ and Cl– ions. CuO is neutral by Cu2+ and O2- ions. FeCl3 is neutral by its Fe3+ and 3Cl– ions. Carbonic acid (H2CO3) is neutral by its two H+ and one CO32- ions.
Use of Roman Numerals:
The Roman numerals are also used as oxidation states but in parentheses with atoms. This practice is necessary for those atoms show variable oxidation states. For example, for ferrous chloride (FeCl2) as iron(II) chloride; because the cation is ferrous (Fe2+). For ferric chloride (FeCl3) as iron(III) chloride; because the cation is ferric (Fe3+). For cupric oxide (CuO), it would be copper(II) oxide; and for cuprous oxide (Cu2O), it would be copper(I) oxide. Usually, the transition metals show multiple oxidation states in complexes. The complexes are the compounds of transition metals in a very simple term.
Redox:
The term is a combination of two words, reduction and oxidation, i.e., red+ox. So, reduction and oxidation reactions are studied under this term. Because, these two half reactions go simultaneously, thus, studied together.
Definitions of Reduction & Oxidation:
- The reduction can be defined as, “the loss of oxygen, gain of hydrogen, gain of electron(s), or decrease in oxidation state during redox reactions”.
- The oxidation can be defined as, “the gain of oxygen, loss of hydrogen, loss of electron(s), or increase in oxidation state during redox reactions”.
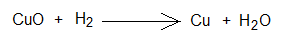
- In this example, Cu2+ of CuO loses oxygen, so, reduction taking place in copper cation of cupric oxide.
- The oxidation state of copper in cupric oxide is +2. So, it gains 2 electrons and reduced to Cu-neutral atom. It is reduction in oxidation state from +2 to zero.
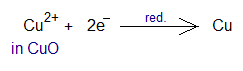
- Hydrogen molecule splits into neutral hydrogen atoms which then lose one electron and increase their oxidation state zero to +1. Then they gain oxygen (oxide ion) to form water. This is all oxidation.
Following diagram shows the increase or decrease of oxidation state by means of oxidation or reduction. Oxidation moves towards right side on the number line, while reduction moves opposite.
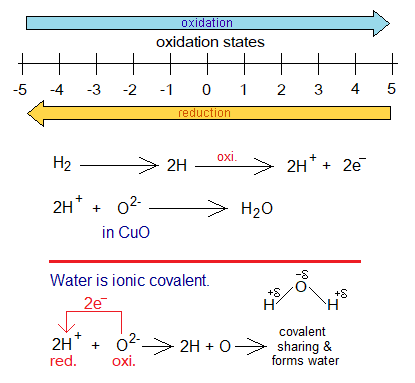
- The electrons released during oxidation are used to reduce cations. The number of electrons released by oxidation is equal to the number of electrons gained by reduction.
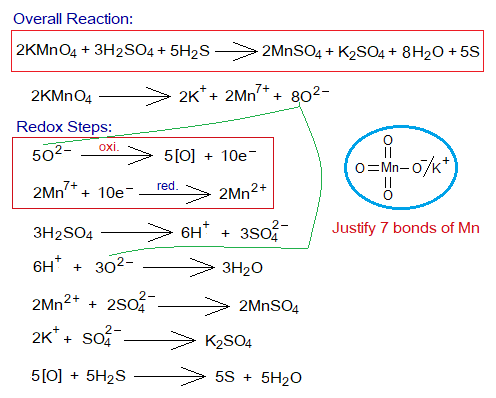
Redox Reaction of KMnO4 with H2S:
The aqueous solution of potassium permanganate [also potassium manganate (VII) KMnO4] is purple (very dark pink) in colour. It shows redox chemical reactions with reducing agents like hydrogen sulfide, oxalic acid etc. in the presence of H2SO4 solution.
- By redox reactions the colour of KMnO4 solution disappears.
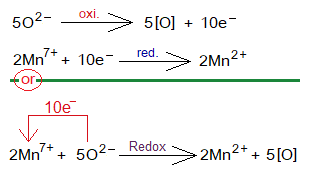
- 2 moles of KMnO4 splits into 2 moles of K+, 2 moles of Mn7+ and 8 moles O2- ions. [2KMnO4=2K++2Mn7++8O2-].
- Five moles of oxide ions show oxidation by 10 moles of electrons in a total (each by 2 moles of electrons). [5O-2=5O+10e–].
- These 10 moles of electrons are then used to reduce 2 moles of manganese heptavalent cations (Mn7+)into oxidation number divalent cations (Mn2+). [2Mn7++10e–=2Mn2+].
Knowledge in Advance:
In-detail step-by-step reactions are shown below:
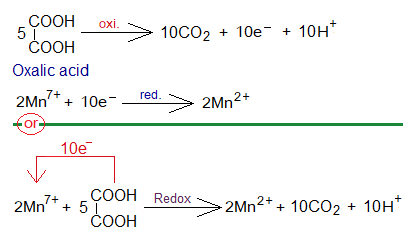
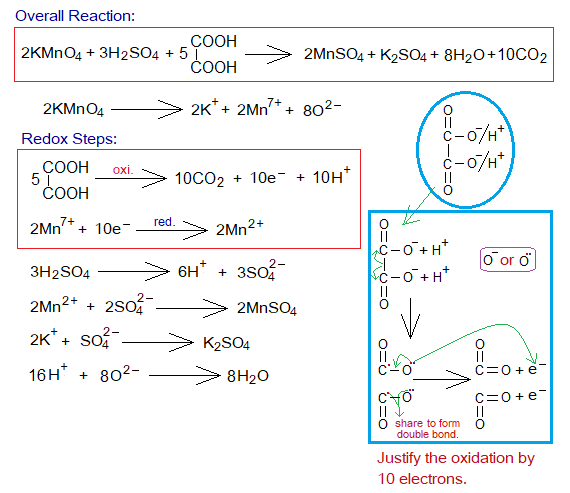
Redox Reaction of KMnO4 with H2C2O4:
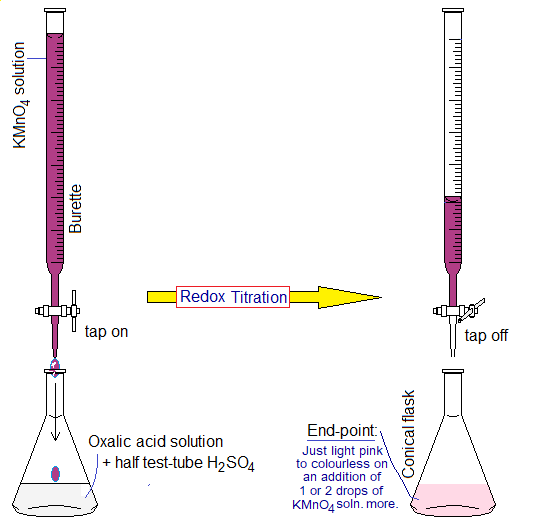
Potassium permanganate also shows redox-titration with oxalic acid (H2C2O4) in the presence of H2SO4 solution.
- By redox reactions the colour of KMnO4 solution disappears.
- KMnO4 splits into 2 moles of K+, 2 moles of Mn7+ and 8 moles O2- ions, as above.
- Five moles of oxalic acid form 10 moles of CO2, 10 moles of H+ ions, and show oxidation by 10 moles of electrons.
- These 10 moles of electrons are then used to reduce 2 moles of manganese Mn7+ into Mn2+, as above.
Knowledge in Advance:
In-detail step-by-step reactions are shown below:
Redox Reactions of Aqueous KI with K2Cr2O7:
Chemical reaction between aqueous solutions of potassium iodide (colourless) and potassium dichromate (orange-red) shows redox mechanism in the presence of H2SO4 solution.
- K2Cr2O7 splits into 2 moles K+, 2 moles Cr6+ and 7 moles of O2- ions.
- Six moles of KI split into 6 moles of K+, and 6 moles of iodide ions (I–).
- Six moles of I– show oxidation by 6 moles of electrons (1 electron by 1 iodide ion). The oxidation number of iodide ion (-1) increases to zero (iodine atom). These iodine atoms then share their electrons of valence shell to form iodine molecule (I2).
- These 6 moles electrons are then used to reduce 2 moles of chromate hexavalent cations (Cr6+)into oxidation number trivalent cations (Cr3+). 2[Cr6++3e–= Cr3+].
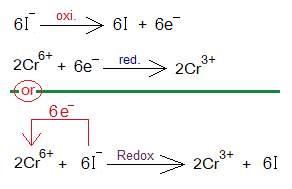
Knowledge in Advance:
In-detail step-by-step reactions are shown below:
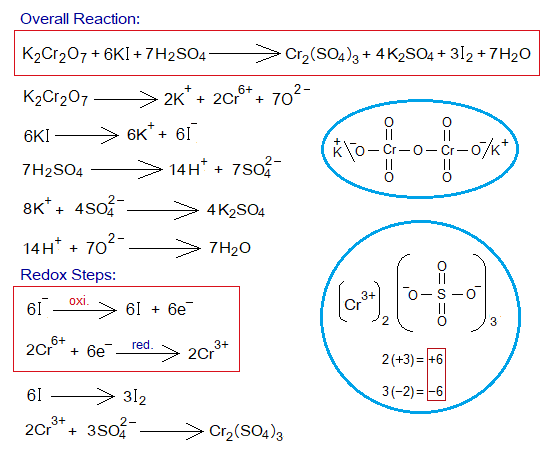
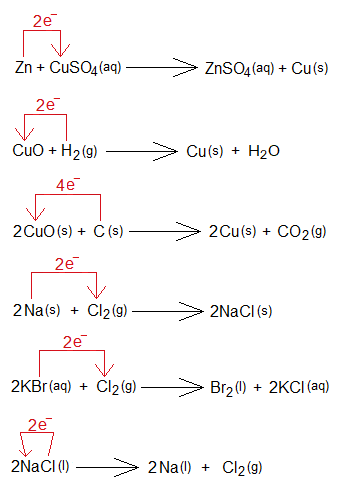
Exercise 1:
By using the following examples, explain oxidation & reduction.
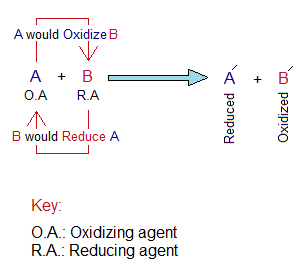
Oxidizing Agent Versus Reducing Agent:
Definitions:
- “A substance that oxidizes the reducing agent, and is reduced by reducing agent” is called oxidizing agent (or oxidant). It needs electron(s) and for that it accepts them from reductant. By the following diagram, the A accepts electrons from B. So, A is an oxidant.
- “A substance that reduces the oxidizing agent, and is oxidized by oxidizing agent” is called reducing agent (or reductant). It is a source of electron(s) that provides them to oxidant. By the following diagram, the B provides electrons to A. So, A is a reductant.
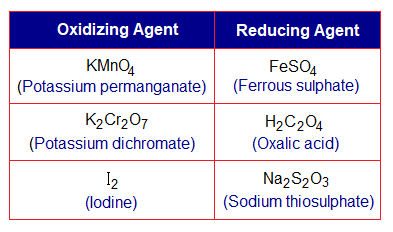
In chemistry laboratory, following are the important reagents used in redox titrations.
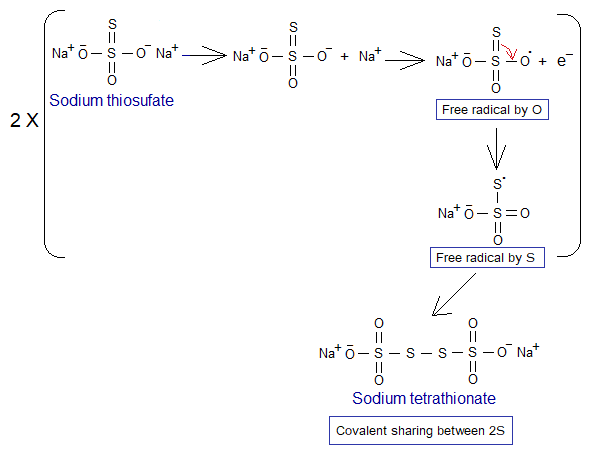
In the following redox example, 1 mole iodine (I2) needs 2 moles electrons to be reduced to 2 moles of iodide ions (I–). These electrons are provided by sodium thiosulfate and resultantly, tetrathionate ions are formed.

Knowledge in Advance:
Exercise 2:
For the following examples, write oxidation and reduction half-reactions with balanced electrons oxidized and used in reduction.